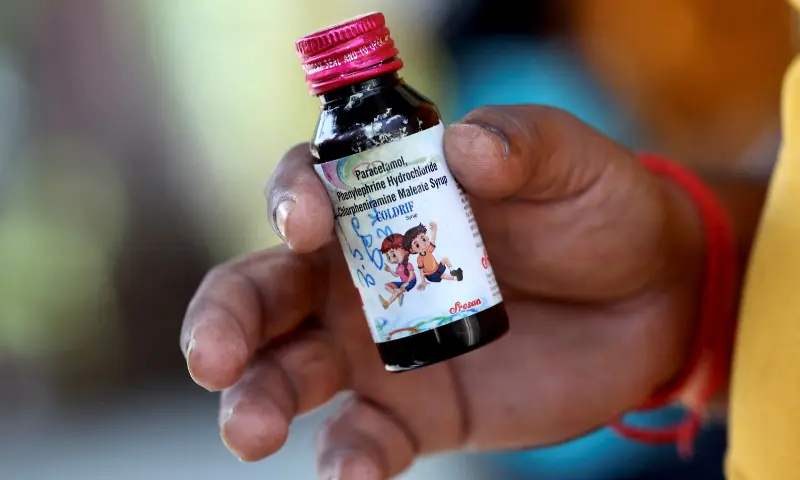
CHHINDWARA: Indian police have arrested the owner of Sresan Pharmaceutical Manufacturer, the cough syrup company linked to the deaths of 19 children in Madhya Pradesh.
The children, all under five years of age, died in the past month in the state’s Chhindwara city, after consuming cough medicine containing toxic diethylene glycol in quantities nearly 500 times the permissible limit.
The deaths were all linked to Sresan Pharma’s Coldrif syrup, which has been banned in several parts of India after a test confirmed the presence of the chemical last Thursday.
S. Ranganathan, owner of the Tamil Nadu state-based company that manufactured the syrup, was arrested on Wednesday in Chennai where he will appear in court before being moved to Chhindwara, a police official said.
Local drug authorities have stepped up efforts to curb the circulation of contaminated medicine, testing random samples of cough syrups and making door-to-door calls to retrieve Coldrif bottles. “We (have) got 30-40 bottles (through this means)…we (also) recalled some from retailers and stockists,” district administrator Harendra Narayan said, adding that 594 bottles of the syrup were sold to pharmaceutical stockists in the region over the last six weeks.
By law, Indian drugmakers must test each batch of raw materials and the final product.
Exports of cough syrup require another layer of tests at government-mandated laboratories since 2023, after the deaths of over 10 children in Gambia, Uzbekistan and Cameroon were linked to Indian syrups.
The World Health Organisation has said the recent case highlights a “regulatory gap” in India’s screening of medicines and warned that some exports could have been made unofficially.
Published in Dawn, October 10th, 2025