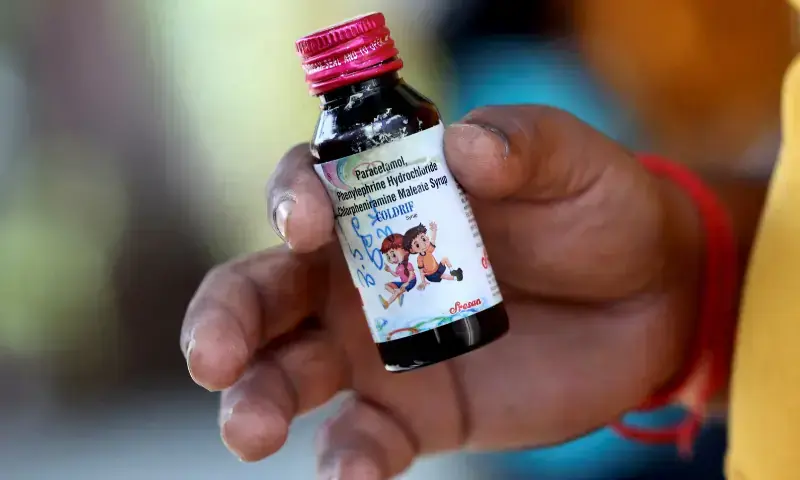
India has more work to do in halting sales of toxic cough syrup, despite some progress, a World Health Organisation (WHO) official told Reuters, after at least 24 children died following consumption of a domestically-made medicine.
The children died after taking the Coldrif cough medicine made by Sresan Pharma, which tests showed contained the toxin diethylene glycol in quantities nearly 500 times the permissible limit.
They came just two years after global pledges to tighten the system following the deaths of at least 300 children around the world linked to similar toxins in syrup-based medicines made in India and Indonesia.
But enforcement issues persist, the WHO said.
“They have made some strides,” said the official, Rutendo Kuwana, referring to a new Indian rule requiring medicine to be tested for contaminants such diethylene and ethylene glycol before export.
However, no such rule exists for syrups sold locally — a “regulatory gap” the WHO has flagged.
“It’s a work in progress,” added Kuwana, the WHO team lead for incidents involving substandard and falsified medicines.
“There’s a lot that needs to be done. It’s a big market, with tens of thousands of manufacturers and many states to deal with.”
Last week, Reuters reported that India plans to scrap its export rule once companies upgrade their facilities to international standards by a year-end deadline.
India’s health ministry and the Central Drugs Standard Control Organisation (CDSCO), the federal pharmaceuticals regulator, did not return requests for comment on the plans.
Representatives of Sresan Pharma did not respond to repeated telephone calls.
A WHO spokesperson said by email on Monday that the agency welcomed all steps to improve medicine quality.
However, in response to a question about dropping the export tests, the spokesperson said medicines, including raw materials, should be tested throughout the production process, not just at the end.
By law, Indian drugmakers must test each batch of raw materials and the final product, but CDSCO said in October some firms had not been doing this.
Kuwana said countries, including Pakistan had attended WHO training, on testing for the toxins, and a new, cheaper test had been developed.
Representatives from India did not attend but it has confirmed to the WHO that it is using the method, he added.
There has also been a lack of accountability for the previous incident, Kuwana said, which he described as a “big disappointment”. Despite the deaths abroad, there is no record of anyone being jailed in India.
“This is not just selling fake shoes,” said Kuwana. “This is a moral issue but if you don’t follow the rules, it’s criminal, because it has devastating consequences.”